THE UNMET NEED
Previously out-of-reach insights about complement
Complement has emerged as a key factor in many conditions.
view_timeline
Complement is an evolutionarily-old cascade of fluid-phase and membrane-associated proteins designed to trigger, amplify, and regulate immunity and inflammation. This central role in the immune network highlights the multiple ways complement dysregulation can drive or modulate immune-mediated diseases.
Immune proteins in general, and complement proteins specifically, are highly labile and unstable, making them difficult to assess clinically. Thus, only limited complement tests are available on large scale and the interpretation and clinical value of those results are restricted.
Safe and effective cytokine and complement-targeted therapies are available. However, their use has been limited by the absence of clinically reliable diagnostics. Delivering actionable diagnostics will provide the insights to unlock the full potential of these immune-based therapies.
By measuring specific elements in the complement cascade quickly, accurately and reliably, we can open the door to a deeper understanding of the role of complement, inform existing treatment decisions for better outcomes, and pave the way for the next generation of therapies.
help_center
Why is complement so hard to measure?
Complement is designed to respond rapidly to danger signals. The same characteristics that allow it to recognize and self-activate in response to danger make it prone to non-specific activation. Thus, once a biological sample has been collected, the complement levels begin to change rapidly. To make things more complicated, the rate and extent of activation is highly variable. There is no conversion factor to reliably correct for these aberrations.
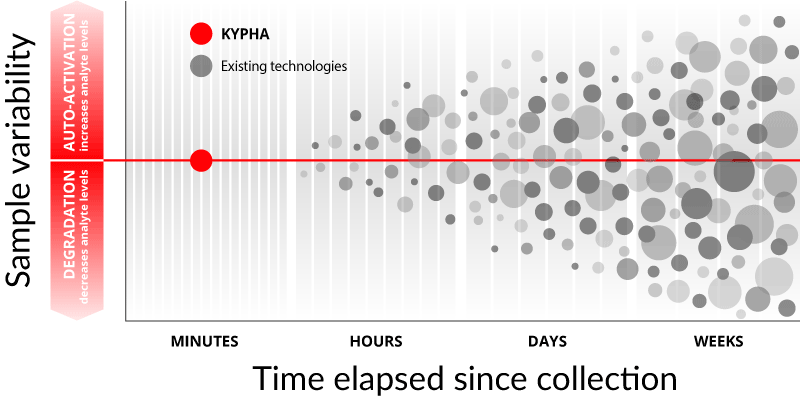
Historically the turn-around time for complement samples has taken days or even weeks, and the impact of degradation and auto-activation is felt almost immediately. Consequently complement protein results bear an uncertain relationship to the in vivo value at time of collection and make current complement testing error-prone and largely unreliable.
Kypha’s technology gives real-time insight into complement proteins – as well as cytokines, chemokines and other biologically labile signals. By enabling measurement of these samples within minutes, we can assess an individual’s immune status with a far higher degree of accuracy and reproducibility. And because it is embedded into our detection technology, this information can be readily available at the point of care.
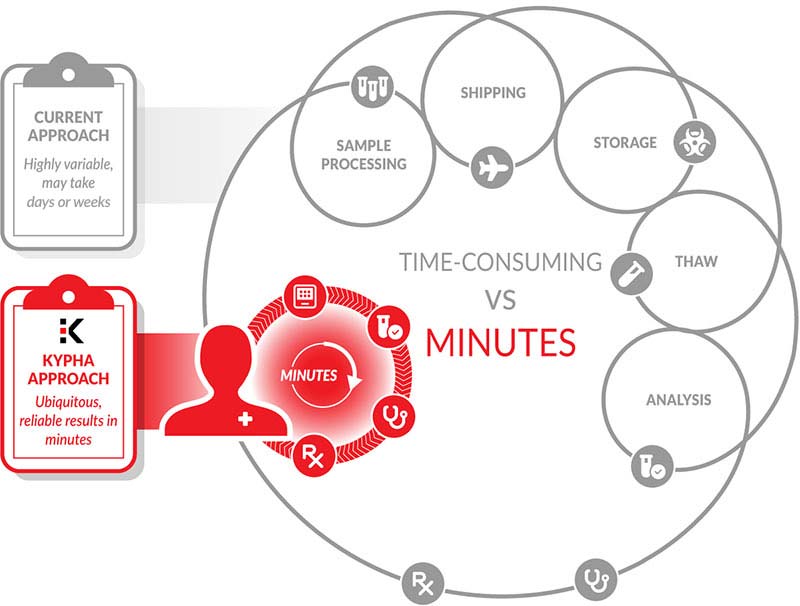
Addressing practical challenges with complement measurement
Kypha’s technology allows the development of real-time assays for highly-labile immune signaling proteins including complement and many cytokines.
analytics
Embedded in our platform technology is the ability to develop panels for the simultaneous measurement of up to multiple (up to 24) distinct and diverse proteins, and to provide the results within minutes from freshly collected and unprocessed patient samples.